Optical imaging is a valuable tool in biology to visualize different structures from sub-cellular level to whole tissues and organs. Nowadays mostly fluorescence imaging is used to get 2D or 3D visual information of biological specimens. This technique has a resolution up to a theoretical limit of roughly 200 nm. Fluorescent molecule dyes can be applied to stain different parts and structures of the biological system, or autofluorescence of the sample can be used directly for imaging, as most of the biological tissues emit fluorescence per se.
Fluorescence
Fluorescence is a property of materials where atoms or molecules emit light due to excitation and relaxation of electrons. To understand the phenomenon, first we have to look at light-material interactions and discuss the very basics of quantum theory. From quantum theory we know that in atoms and molecules, electrons are localized and can only occupy distinct energy levels (states). For organic molecules, numerous different energy levels are exist due to the high number of electrons and bonds. Normally electrons stay in the lowest energy level (ground state, S0 in the figure). If a specific color of light (energy) is shined onto the material, an electron in the ground state may absorb it and get transferred to an excited state with a higher energy level (S1 in the figure).
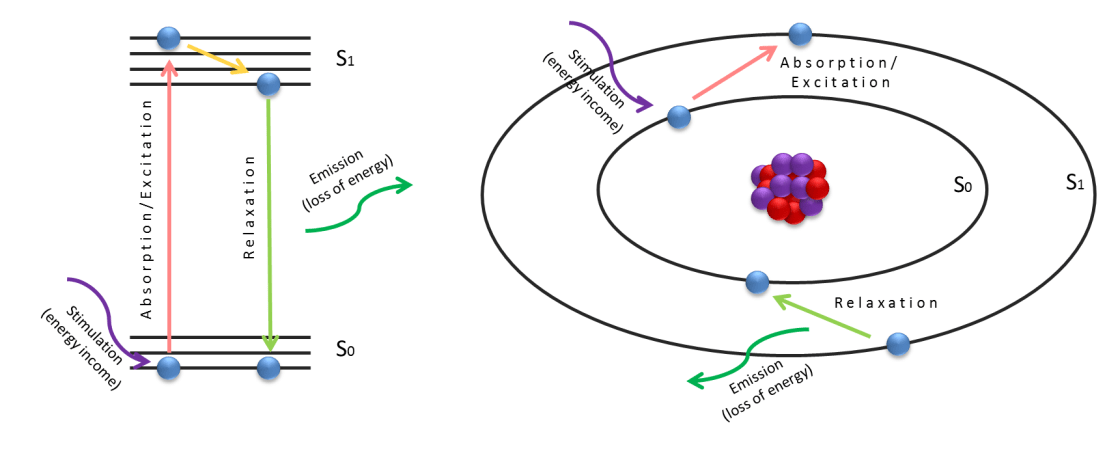
The electron then “immediately” falls back to the ground state as it cannot occupy a higher energy level for an increased period of time. The energy difference between the excited and ground states is emitted out as a photon in the relaxation process (green arrow in the figure). The color of the photon is representing the energy difference of S1-S0 and it’s material dependent as the energy level of the states are variate in different atoms or molecules. Therefore every atom’s and molecule’s emission is a signature, and with that, the atom or molecule can be identified (for more info on this, see spectroscopy). If the emission’s energy falls into the visible spectrum of light, we can directly see it with our eye or can be visualized with camera- or detector-based imaging systems. In molecules contrary to atoms, the color of the absorbed photon and the emitted photon is different, the emitted photon’s color is always red-shifted from the excitation source’s color due to the energy dissipation in the excited states (Stokes-shift, yellow arrow in the figure). Therefore the emission light can be separated by the excitation light with optical filters, and fluorescence-based imaging systems can be built to acquire only the emitted light of the different specimens.
For a gallery of fluorescence images, click here.
Fluorescence-based imaging systems
Fluorescence microscopes use a light source (incandescent, led or laser) to excite the fluorescent molecules in the specimen and collect their emission via an optical system. The emission is specific to the used fluorescent dyes (or autofluorescence from the sample per se), therefore different fluorescent dyes can be used to label various biological structures and the whole specimen can be visualized with multi-color imaging.

The figure above shows a laser scanning microscope setup, where a scanning laser beam (green) is exciting the sample, and the emission (red) is collected from the focused 2D plane. By moving the sample position up or down, multiple 2D planes of the sample can be visualized, and a full 3D dataset can be rendered.
All lens-based optical systems (microscpes, telescopes) are diffraction limited. This means that a theoretical resolution is introduced in all optical microscopes, limiting the smallest visible structures to roughly 200nm.
For a gallery of fluorescence microscopy images, click here.
